
Tina Bhardwaj explores nanotechnology, a new development in dentistry with the potential to create more durable restorations.
Imagine a world where dental cavities can heal themselves before they cause pain. With the rise of nanotechnology, this vision becomes a reality. Nanotechnology is the manipulation of matter at the atomic scale, and has transformed several areas of medicine already. In dentistry, its application promises the creation of more durable, biologically compatible materials, transforming both preventive and restorative care.
While preventive dentistry solely focuses on patient compliance alongside fluoride based interventions restorative materials continue to face challenges regarding their aesthetics, biocompatibility and mechanical resilience (Dipalma et al, 2024).
Nanotechnology can work to offer solutions to both of these issues by enhancing material performance, and introducing nano sized tri-calcium phosphate (TMP) in fluoride toothpaste (Nagata et al, 2023), thereby enhancing remineralising potential for patients and reducing early carious lesions.
Despite the potential, nanotechnology faces significant barriers to clinical usage due to high production costs, ongoing ethical debates and safety concerns.
This article intends to explore how nanotechnology is increasingly influencing both preventive and restorative dentistry by evaluating current uses and projecting the future of nanotechnology. I will focus on three core areas of dentistry: endodontics, periodontal disease and dental materials.
Methods
This paper is based on structured literature reviews of current scientific evidence exploring nanotechnology in endodontic applications, periodontal therapy and nano-composites. The sources used were gathered from peer reviewed journals, using Pubmed and Sciencedirect.
The key studies examined include those on nano-hydroxyapatite remineralisation, antibacterial nanoparticles in endodontics and nano-drug delivery in periodontal regeneration.The sources used had a key focus on systematic reviews and experimental data in order to assess the current impact with any potential development of nanotechnology in dentistry.
Results
Nanotechnology has begun to reinvent the quality and longevity of restorative materials in dentistry. Although traditional composites have shown functionality, they fall short by having a shorter lifespan depending on a number of factors (Wellness Dental, 2024).
However, nano-composites as studied by Mitra et al (2003) and Ilie et al (2009), show outstanding improvements in surface smoothness, mechanical strength and fracture resistance using non-agglomerated silica nanoparticles, nanoclusters and nano-hybrid composites. Ultimately, this produces restorations which are more durable and polishable due to filler size, allowing better marginal sealing and resistance to wear overtime.
In addition to structural improvements, nano-hydroxyapatite has recently materialised as a powerful incorporation to fluoride toothpaste. Daas, Badr and Osman (2018) discovered nano-HAP (hydroxyapatite) paste showed promising long term protection in terms of surface depositions in initial caries like lesions thus maintaining a smooth surface. Based on these findings, nano HAP paste could be recommended as an alternative to remineralising agent with a lower fluoride concentration, beneficial for those at risk of dental fluorosis.
What challenges can arise?
One of the main challenges presented with root canal therapy is the possibility of reinfection due to the failure of complete bacterial elimination of canal systems (Dental Perfections, 2025). Metallic nanoparticles can be seen as effective due to their antimicrobial properties, in particular silver nanoparticles (AgNPs) (Oncu et al, 2021).
AgNPs have been tested as endodontic retrofill materials, irrigation solutions and canal sealers, and studies report on the effectiveness as follows. Lotfi et al (2019) compared the effectiveness of AgNPs against E faecalis, alongside NAOCl in an irrigation solution; 5.25% NaOCl and low-conc AgNPs showed similar bactericidal effects. However, Rodrigues et al (2018) found that AgNP solution eliminates fewer bacteria but dissolves more biofilm compared to chlorhexidine (P<0.05) as an irrigant, thus presenting a limitation.
Treating periodontal disease with nanotechnology
Periodontal disease is characterised by inflammation and infection of the gums, often difficult to treat and stabilise depending on the severity. A recent source by Di Wang et al (2024) offers long term solutions to periodontal disease using nanotechnology. By focusing on inorganic nanoparticle (metal oxide, hydroxyapatite NPs), nanoparticulate composites (films, hydrogels) and organic nanoparticles (Polydopamine (PDA NPs)), these can serve as active agents in killing bacteria, preventing biofilm regrowth and supporting inflammation reduction.
As shown in Figure 2, PDA nanoparticles significantly reduced the expression of cytokines markers TNF-α and IL-1β. When treatment with lipopolysaccharide alone was carried out, there was an increase in cytokine levels, compared to the control. However, co-treatment with PDA NPs led to a drastic reduction in both cytokine mediators, demonstrating the nanoparticle’s potential to suppress inflammation by actively modulating the immune response.
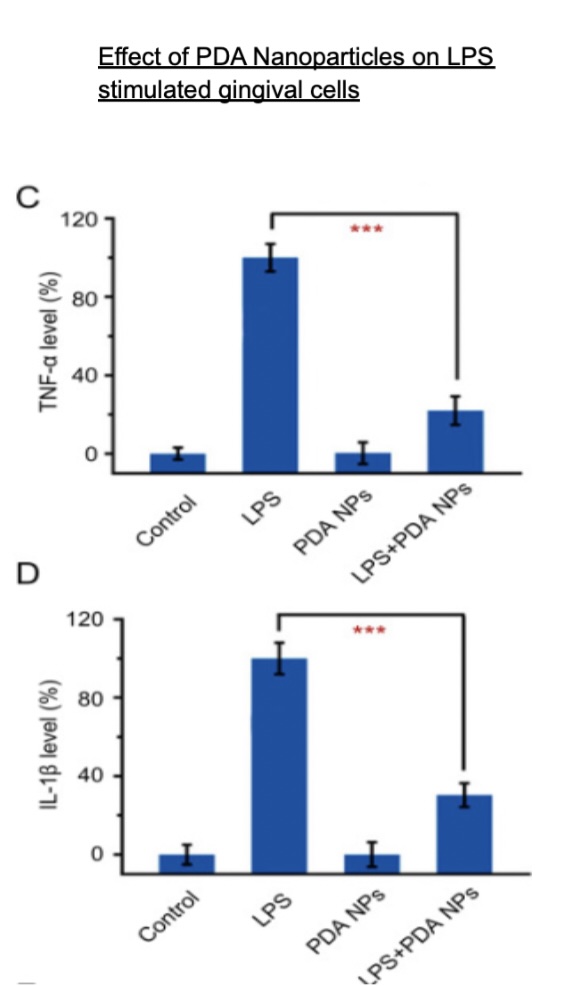
Figure 2: Diagram depicting the anti-inflammatory effects of polydopamine nanoparticles (PDA NPs) on Lipopolysaccharide (LPS) stimulated human gingival epithelial cells (Adapted from Di Wang et al 2024)
Discussion
The emergence of nanotechnology in our modern day society can play an integral role in the future of dentistry, beyond traditional treatments. By enabling stronger and biologically compatible restorative materials which can promote enamel remineralisation, nanotechnology is able to address one of the major challenges in restorative dentistry. In doing so, patient satisfaction and efficiency is improved.
However, many of the studies were limited by in vitro designs and small non-randomised sample sizes, limiting the generalisability of the findings (Mitra et al, 2003). Without having longitudinal clinical evaluation, the real world performance of bio-composites remains questionable.
In endodontics, nanotechnology has presented the opportunity to address challenges in root canal therapies. Focusing on silver nanoparticles (AgNPs), used in sealers and irrigants, they offer the benefits of antimicrobial and anti-biofilm effects allowing a more targeted approach to treatment. However, further clinical evaluation is required to assess long term impacts and patient suitability.
Similarly, in periodontics, the shift towards using various nanoparticle systems have demonstrated the potential to suppress inflammation, and promote an antibacterial function. As Wang et al (2024) stated, these systems can act as both an active agent and delivery system, transporting targeted therapeutic molecules deep into the tissue (Puri and Puri, 2013).
Figure 2 highlights the potential in modulating immune responses in periodontal therapy. Despite these claims, current research is pre-clinical and requires further funding alongside consent for human trials to fully weigh up their impacts.
Conclusion
Nanotechnology can redefine dentistry as we know it. By enabling treatments from offering stronger dental materials, targeted treatment for periodontal disease and the future scope of regenerating pulpal tissue within endodontics, the clinical benefits are beyond traditional.
While ethical, financial and real world application challenges remain, the paradigm shift towards regenerative dentistry is already on its way. With continued research into the safety and clinical impact of nanotechnology, the future of dentistry could already be in our hands.
References
- Dipalma, G, Alessio Danilo Inchingolo, Guglielmo, M, Morolla, R, Palumbo, I, Riccaldo, L, Mancini, A, Palermo, A, Malcangi, G, Angelo Michele Inchingolo and Inchingolo, F (2024). Nanotechnology and its application in dentistry: a systematic review of recent advances and innovations. Journal of Clinical Medicine, [online] 13(17), pp.5268–5268. doi:https://doi.org/10.3390/jcm13175268.
- Nagata, ME, Delbem, ACB, Baez-Quintero, L, Danelon, M, Sampaio, C, Monteiro, DR, Wiegand, A and Pessan, JP (2023). Effect of fluoride gels with nano-sized sodium trimetaphosphate on the in vitro remineralization of caries lesions. Journal of Applied Oral Science, 31. doi:https://doi.org/10.1590/1678-7757-2023-0115.
- Cosmetic Dentist Surprise | Best Dentist in Mesa, Tucson Dentist. (2024). Understanding the Risks: The Hidden Dangers of Composite Fillings. [online] Available at: https://www.mywellnessdental.com/blog/dangers-of-composite-fillings/.
- Mitra, SB, WU, D, and Holmes, BN (2003). An application of nanotechnology in advanced dental materials. The Journal of the American Dental Association, [online] 134(10), pp.1382–1390. doi:https://doi.org/10.14219/jada.archive.2003.0054.
- Ilie, N and Hickel, R (2009). Investigations on mechanical behaviour of dental composites. Clinical Oral Investigations, 13(4), pp.427–438. doi:https://doi.org/10.1007/s00784-009-0258-4.
- Daas, I, Badr, S and Osman, E (2018). Comparison between Fluoride and Nano-hydroxyapatite in Remineralizing Initial Enamel Lesion: An in vitro Study. The Journal of Contemporary Dental Practice, [online] 19(3), pp.306–312.
- www.dentalperfections.co.uk. (ND). Root Canal Treatment Side Effects, Risks and Complications. [online] Available at: https://www.dentalperfections.co.uk/root-canal-risks-hampstead.html (Accessed 2025).
- Oncu, A, Huang, Y, Amasya, G, Sevimay, FS, Orhan, K and Celikten, B (2021). Silver nanoparticles in endodontics: recent developments and applications. Restorative Dentistry & Endodontics, 46(3). doi:https://doi.org/10.5395/rde.2021.46.e38.
- Lotfi, M, S Vosoughhosseini, B Ranjkesh, Khani, S, M Saghiri and Zand, V (2019). Antimicrobial efficacy of nanosilver, sodium hypochlorite and chlorhexidine gluconate against Enterococcus faecalis. African Journal of Biotechnology, [online] 10(35), pp.6799–6803. Available at: https://www.ajol.info/index.php/ajb/article/view/94676 (Accessed 20 April 2025).
- Rodrigues, CT, de Andrade, FB, de Vasconcelos, LRSM., Midena, RZ, Pereira, TC, Kuga, MC, Duarte, MAH and Bernardineli, N (2018). Antibacterial properties of silver nanoparticles as a root canal irrigant against Enterococcus faecalis biofilm and infected dentinal tubules. International Endodontic Journal, 51(8), pp.901–911. doi:https://doi.org/10.1111/iej.12904.
- Wang, D, Li, Q, Xiao, C, Wang, H and Dong, S (2024). Nanoparticles in periodontitis therapy: a review of the current situation. International Journal of Nanomedicine, Volume 19, pp.6857–6893. doi:https://doi.org/10.2147/ijn.s465089.
- Puri, K and Puri, N (2013). Local drug delivery agents as adjuncts to endodontic and periodontal therapy. Journal of Medicine and Life, [online] 6(4), p.414. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC4034307/ (Accessed 20 April 2025).


