RevBio secures FDA approval to begin dental clinical trial for regenerative bone adhesive
RevBio has received approval from the US Food and Drug Administration (FDA) to initiate a pilot dental clinical trial evaluating the safety and efficacy of
RevBio has received approval from the US Food and Drug Administration (FDA) to initiate a pilot dental clinical trial evaluating the safety and efficacy of

In today’s busy life, patients may have trouble coordinating their work and home schedules when treatment requires multiple appointments. Typically, when restoration of teeth is
K Line Europe, the leading OEM Clear Aligners manufacturer in Europe, has recently received FDA registration to introduce its highly popular K Clear Aligners in

The U.S. Food and Drug Administration has provided an at-a-glance summary of news from around the agency: U.S. Food and Drug Administration (FDA) logo (PRNewsfoto/FDA)

Vivos Therapeutics, Inc. (the “Company” or “Vivos”) (NASDAQ: VVOS), a medical technology company focused on developing and commercializing innovative diagnostic and treatment modalities for patients

When Dr. Édouard Lanoiselée met a patient who needed an old amalgam inlay restoration changed with a tooth color restoration, the typical process was straightforward.
Henry Quach explores the potential impact of delivering the COVID-19 vaccines 12 weeks apart instead of 21 days apart. The UK government is now delivering
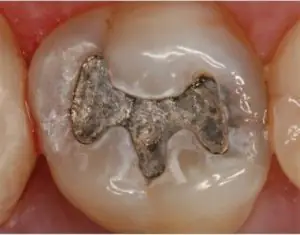
The Food and Drug Administration (FDA) now recommends that people who belong to certain high-risk groups avoid getting dental amalgam whenever possible and appropriate due

The Food and Drug Administration (FDA) needs you! Its Medical Devices Advisory Committee (MDAC) Dental Products Panel is looking for dentists, engineers, and scientists with

The United States Food and Drug Administration (FDA) has proposed a rule that would reduce the allowable level of fluoride in bottled water from 0.8
The Food and Drug Administration (FDA) has accepted a pre-market approval (PMA) application from CALCIVIS for its imaging system for substantive review. The imaging system
The US Food and Drug Administration (FDA) has cleared Neocis to market Yomi, a robotic system that guides surgical instruments during dental implant procedures. It
