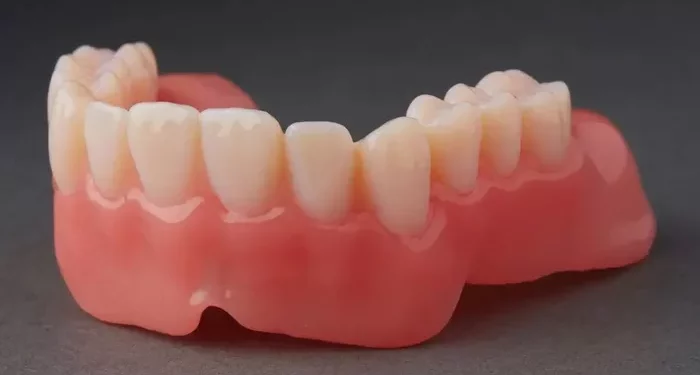
Seoul, South Korea – Graphy Inc. (KOSDAQ: 318060) has secured U.S. FDA 510(k) clearance for its TERA HARZ Hard Denture resin series, accelerating its push into the $40 billion global prosthetic materials market.
The clearance covers eight TERA HARZ models—THD-C-500, THD-C-1000, THD-LP-500, THD-LP-1000, THD-P-500, THD-P-1000, THD-CP-500, and THD-CP-1000—engineered for high strength, durability, and flexibility, making them suitable for 3D-printed dentures.
Graphy’s previous approvals include resins for permanent crowns, bridges, shape memory aligners, surgical guides, flexible dentures, and mouthguards, giving the company a broad footprint in dental 3D printing.
With this latest clearance, Graphy now holds 58 FDA 510(k) approvals and 37 CE certifications, representing one of Korea’s strongest regulatory records in dental 3D printing.
The company’s Shape Memory Aligner technology—world-first in 3D printing clear aligners—has expanded its material portfolio across orthodontics, prosthetics, dentures, and sports guards.
Graphy said the new denture resins enable dental labs to move from traditional hand-crafted workflows to fully digital production, improving precision and efficiency. The company also provides integrated solutions combining 3D printers, software, and validated production protocols for dental clinics and labs.
The global orthodontics market is projected to grow from $70 billion in 2023 to $110 billion by 2030, with North America accounting for 45% of demand. FDA clearance further validates Graphy’s technology and strengthens the company’s competitiveness in international markets.
Graphy plans to expand its U.S. distribution network, deepen partnerships with clinics and laboratories, and engage with global manufacturers.
The company is also promoting its Shape Memory Aligner and 3D printing materials at major international events, including meetings in Taiwan, Dubai, and the United States, aiming to secure new partners and grow its overseas sales pipeline.